Sign up for FlowVella
Sign up with FacebookAlready have an account? Sign in now
By registering you are agreeing to our
Terms of Service
Loading Flow






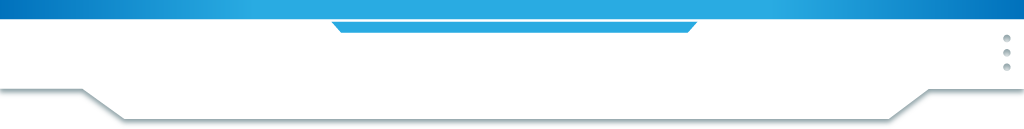

Evidence of Data
Rutherford found that the vast majority of particles were passed straight through the foil. Approximately 1 in 8000 were deflected, leading him to his theory that most of the atom was made up of 'empty space'.