Sign up for FlowVella
Sign up with FacebookAlready have an account? Sign in now
By registering you are agreeing to our
Terms of Service
Loading Flow

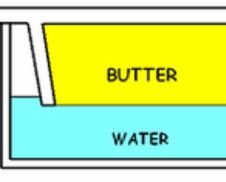
Hydrophilic vs. Hydrophobic
Since the last slide covered the basic meaning, now we can add more detail.
Hydrophilic substances will dissolve in water based substances, but NOT in OIL based substances, and hydrophobic substances will dissolve in oil based substances, but NOT in WATER based substances.
These are called polar salutes and solvents vs. non-polar salutes and solvents.
For example: salt and water


Salt is a polar salute, and water is a polar solvent.
For example: butter and oil.