Sign up for FlowVella
Sign up with FacebookAlready have an account? Sign in now
By registering you are agreeing to our
Terms of Service
Loading Flow



Periodic Trend
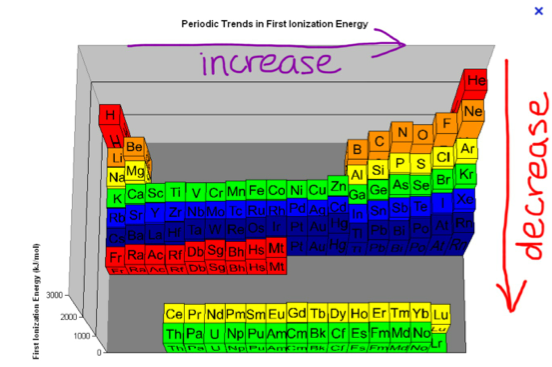



Why Increase?
As you go across the periodic table, the atom gains more electrons, so the proton's force over the electrons is stronger.
Why Decrease?
As you go down the periodic table, the electrons are farther away from the nucleus, so the proton's pull isn't as strong.

