Sign up for FlowVella
Sign up with FacebookAlready have an account? Sign in now
By registering you are agreeing to our
Terms of Service
Loading Flow

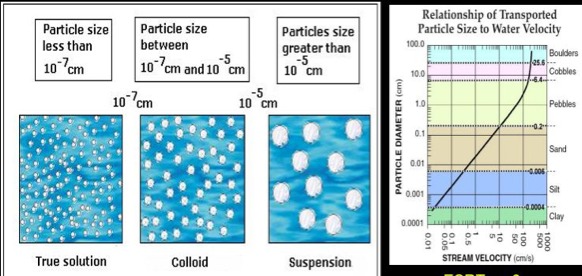
14. Solutions are homogeneous mixtures of components that may be solids, liquids, or gases. Examples are the air we breathe (mixture of gases) and seawater (a mixture of salts, which are solids, and water). The substance present in the greatest amount is the solvent and in the smaller amount is the solute. Most solutions in the body are true solutions containing gases, liquids, and solids dissolved in water. True solutions are usually transparent. Colloids also called emulsions are heterogeneous mixtures that often appear translucent or milky. Although the solute particles are larger than those in true solutions, they still do not settle out. They do scatter light however, and so the path of a light beam shining through colloidal mixture is visible. Colloids have the ability to undergo sol-gel transformations, or to change reversibly from a fluid state to a more solid state. An example would be Jell-O. Suspensions are heterogeneous mixtures with large, often visible solutes that tend to settle out. An example of a suspension is a mixture of sand and water. Blood is another suspension in which living cells are suspended in the fluid portion of blood. If left to stand, the suspended cells will settle out unless mixing, shaking, or in the body, circulation is used to keep them in suspension.
